- Home
- About Us
- Work
- Journal
- Contact
- Panorama maker apple
- Juegos de minions paradise
- Cgsecurity-org testdisk
- Parallels desktop for mac review
- Sookasa free personal plan limits
- Quick menu windows 7
- Valentin imperial riviera maya
- Compounds that join together to form polymers
- Serenity symbol vector
- Umineko when they cry vn
- Setup vpn tracker with sonicwall tz300
- Home
- About Us
- Work
- Journal
- Contact
- Panorama maker apple
- Juegos de minions paradise
- Cgsecurity-org testdisk
- Parallels desktop for mac review
- Sookasa free personal plan limits
- Quick menu windows 7
- Valentin imperial riviera maya
- Compounds that join together to form polymers
- Serenity symbol vector
- Umineko when they cry vn
- Setup vpn tracker with sonicwall tz300

Much of the chemistry that makes life happen involves the making and breaking apart of polymers. Irrespective of the number of monomers, both of these molecules are polymers. While starch, which is another carbohydrate, is made up of numerous glucose units. For example, raffinose, a carbohydrate commonly found in beans and vegetables, is composed of one glucose, one galactose, and one fructose unit. While polymers can contain thousands or millions of monomers, a polymer can contain as few as three monomer molecules.
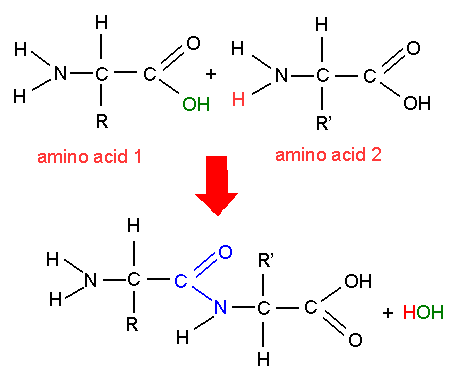
Because polymers are larger structures made up of numerous monomer molecules, polymers are often called macromolecules.

Hundreds of thousands of monomers, even millions of them, can connect or join together to form polymers. Monomers, the building blocks of polymers, can be thought of as a chain like the different types of cars on a train. Nucleotides serve as the monomers for nucleic acids, such as DNA and RNA. Similarly, amino acids in various combinations form protein molecules. For example, glucose units come together to form large cellulose and starch molecules. These components are the building blocks of other molecules. Glucose, amino acids, and nucleotides are examples of monomers. Monomers are thus the molecule-level building blocks of polymers. These molecules, which link together to form polymers, are called monomers. Just as several atoms come together to form an element and elements form molecules, polymers are also formed by the joining of several smaller units, or molecules.
